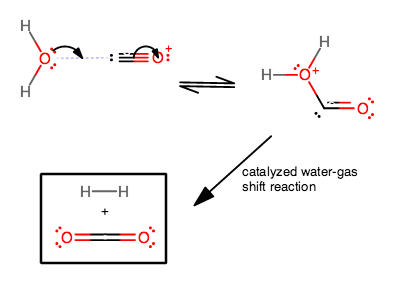
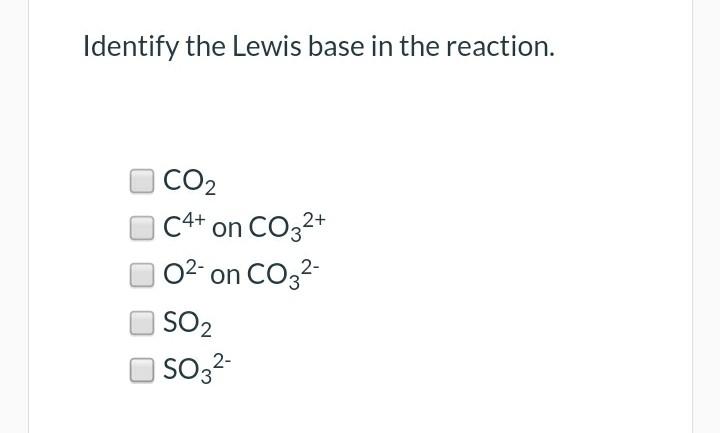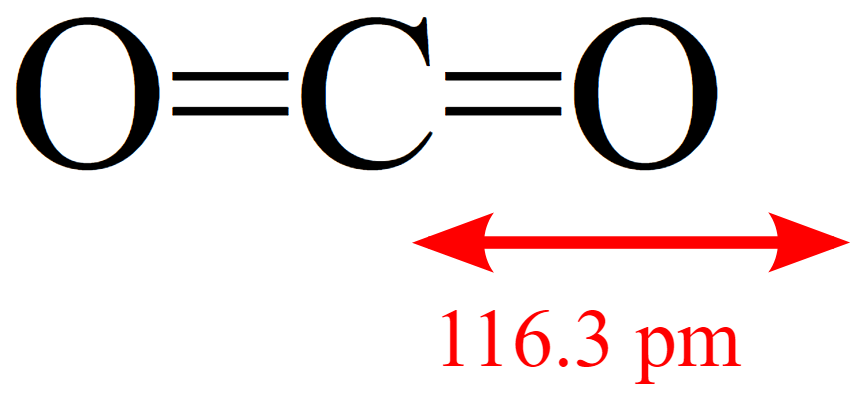Selectively Regulating Lewis Acid–Base Sites in Metal–Organic Frameworks for Achieving Turn‐On/Off of the Catalytic Activity in Different CO2 Reactions - Tian - - Angewandte Chemie International Edition - Wiley Online Library

Role of the Surface Lewis Acid and Base Sites in the Adsorption of CO2 on Titania Nanotubes and Platinized Titania Nanotubes: An in Situ FT-IR Study | The Journal of Physical Chemistry
Minerals | Free Full-Text | CO2 Dipole Moment: A Simple Model and Its Implications for CO2-Rock Interactions

Lewis Acid Strength of Interfacial Metal Sites Drives CH3OH Selectivity and Formation Rates on Cu‐Based CO2 Hydrogenation Catalysts - Noh - 2021 - Angewandte Chemie International Edition - Wiley Online Library

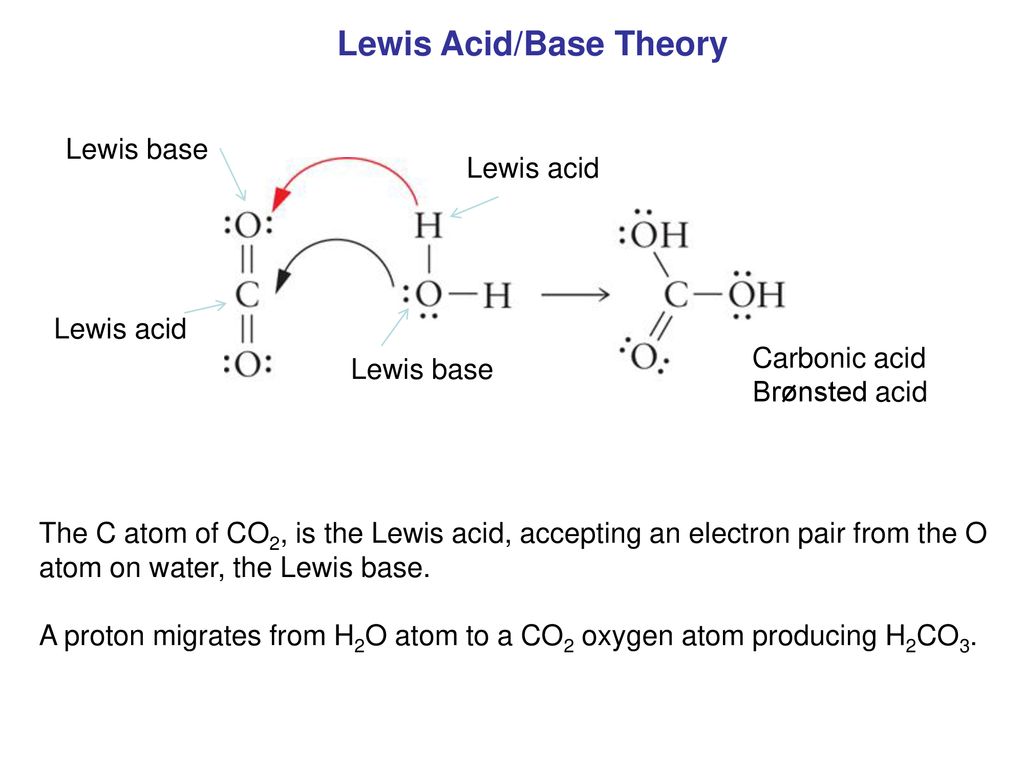
Lewis Acid Enhancement of Proton Induced CO2 Cleavage: Bond Weakening and Ligand Residence Time Effects | Journal of the American Chemical Society
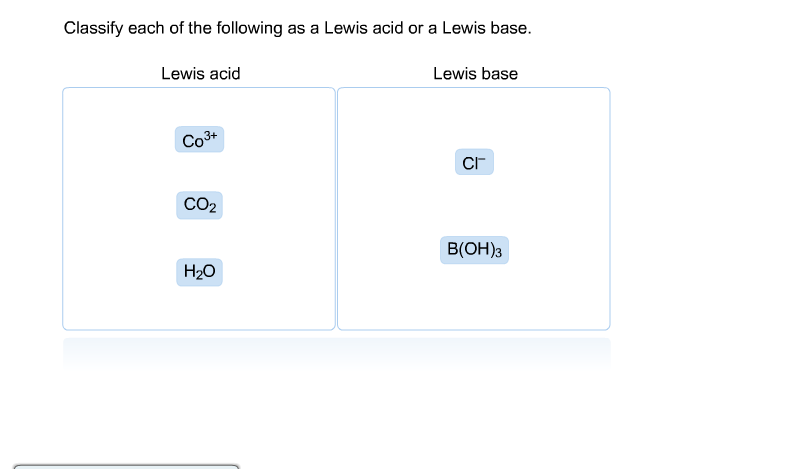
![SOLVED: [ Which of the statements regarding the Lewis acid-base theory is FALSE? ] 1. All Bronsted Lowry bases are also Lewis bases. 2. Non-metal oxides such as CO2 and SO3 are SOLVED: [ Which of the statements regarding the Lewis acid-base theory is FALSE? ] 1. All Bronsted Lowry bases are also Lewis bases. 2. Non-metal oxides such as CO2 and SO3 are](https://cdn.numerade.com/previews/2f9ab192-509b-4230-b66b-21c281c3582f_large.jpg)
SOLVED: [ Which of the statements regarding the Lewis acid-base theory is FALSE? ] 1. All Bronsted Lowry bases are also Lewis bases. 2. Non-metal oxides such as CO2 and SO3 are

Porous metal-organic framework with Lewis acid−base bifunctional sites for high efficient CO2 adsorption and catalytic conversion to cyclic carbonates - ScienceDirect
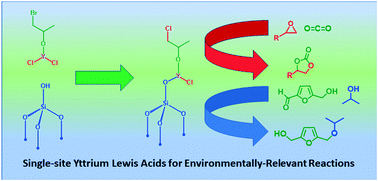
Synthesis of well-defined yttrium-based Lewis acids by capturing a reaction intermediate and catalytic application for cycloaddition of CO2 to epoxides under atmospheric pressure - Catalysis Science & Technology (RSC Publishing)

Selectively Regulating Lewis Acid–Base Sites in Metal–Organic Frameworks for Achieving Turn‐On/Off of the Catalytic Activity in Different CO2 Reactions - Tian - 2022 - Angewandte Chemie International Edition - Wiley Online Library