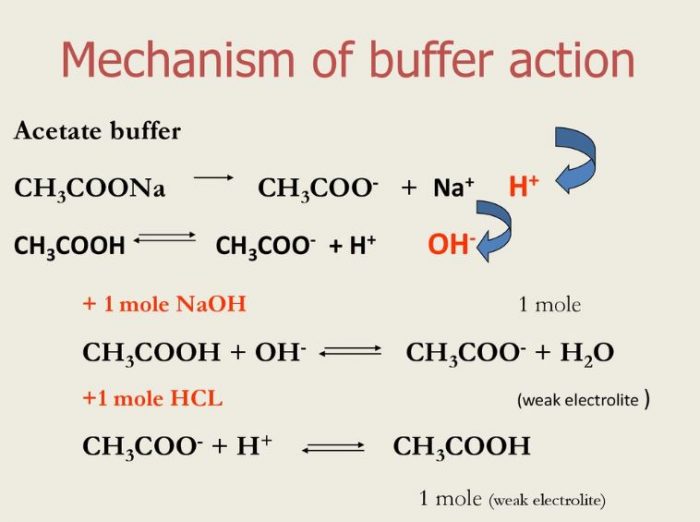
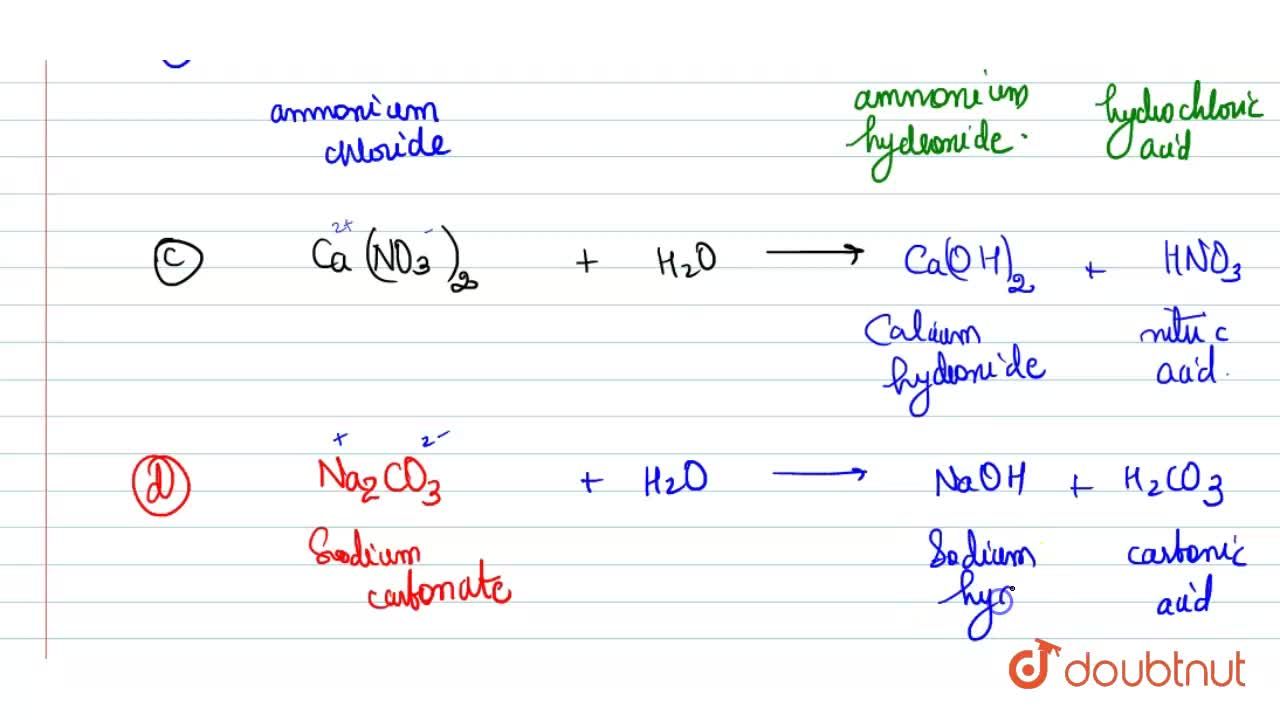
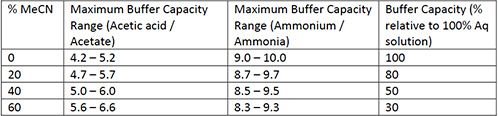
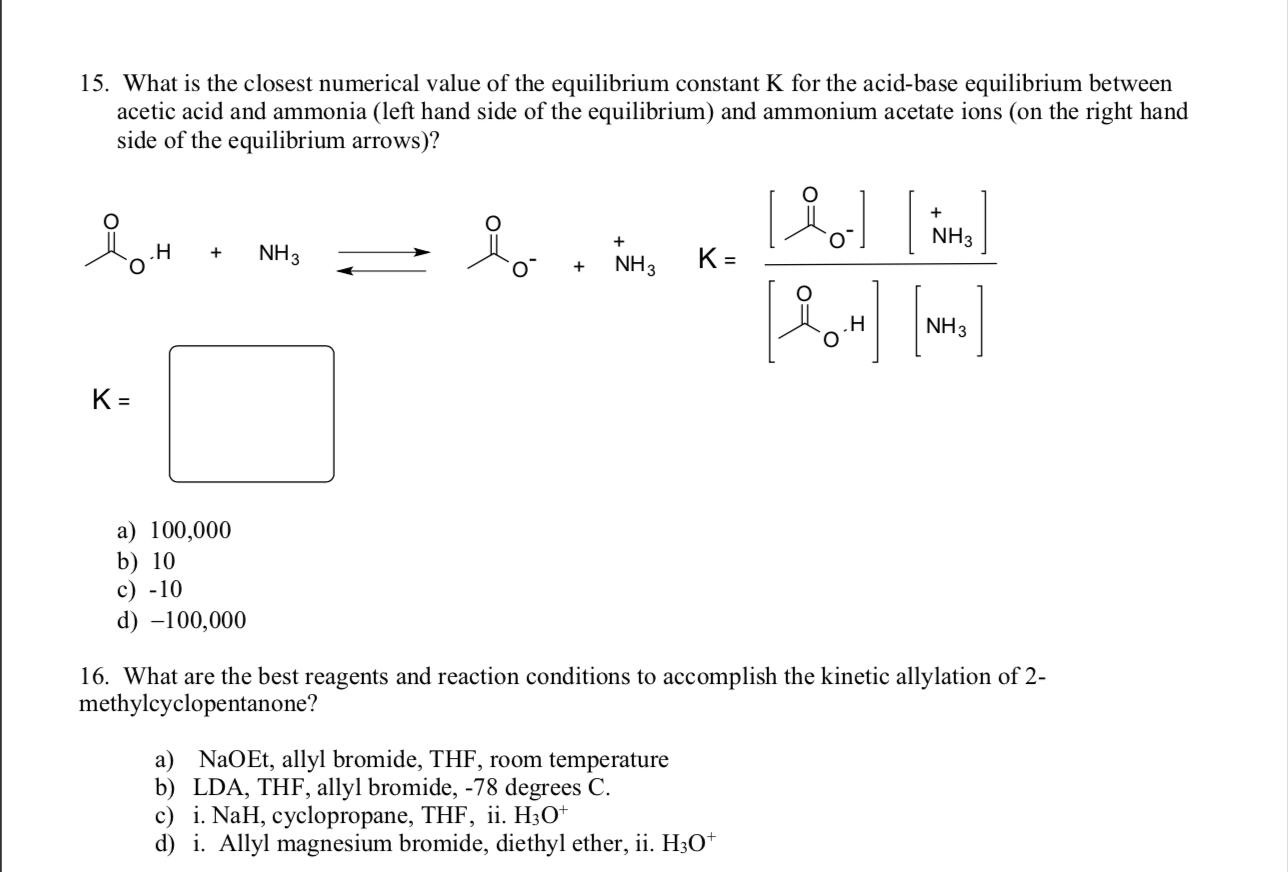
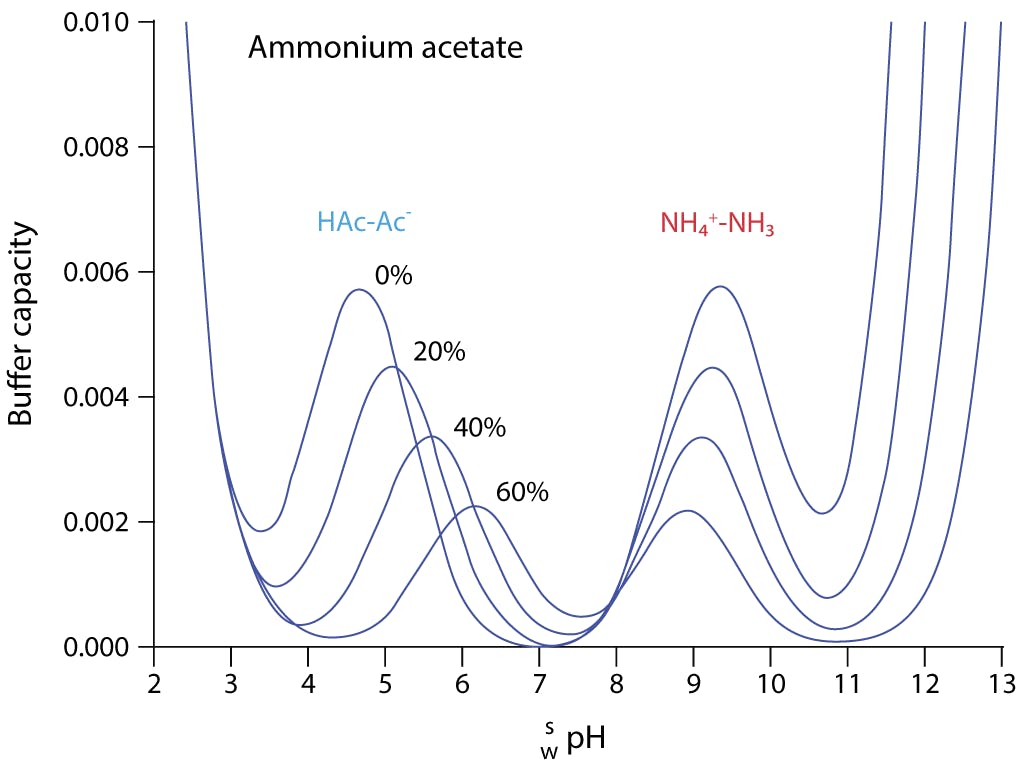
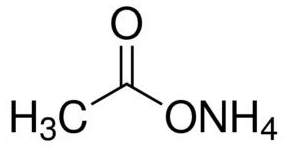
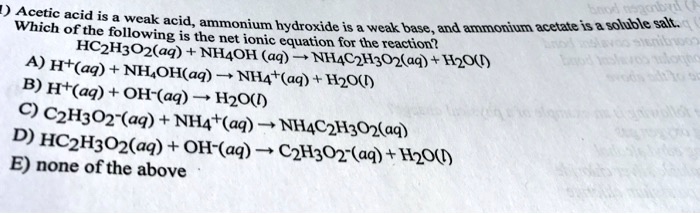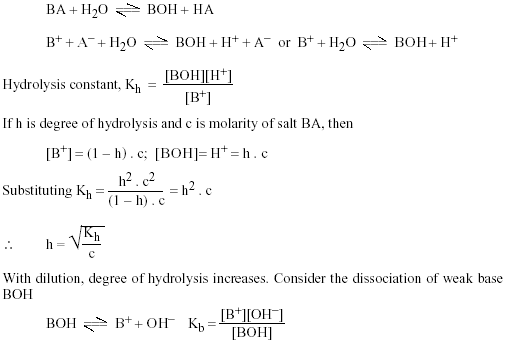pka of acetic acid and pKb of ammonium hydroxide are 4.76 and 4.75 respectively. Calculate the pH of ammonium acetate solution.
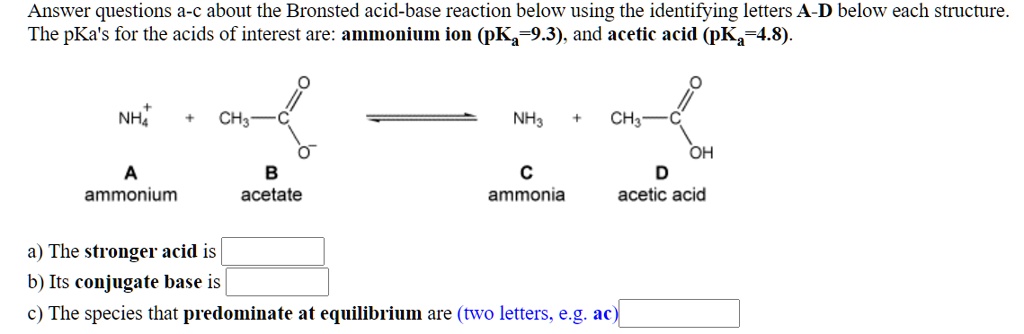
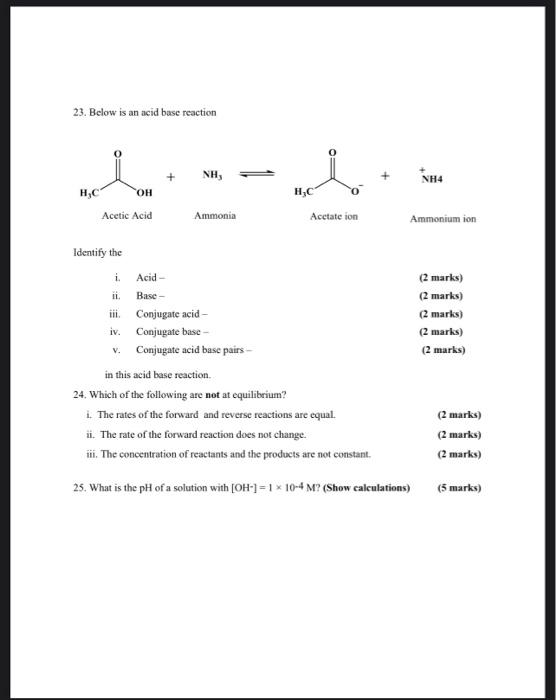
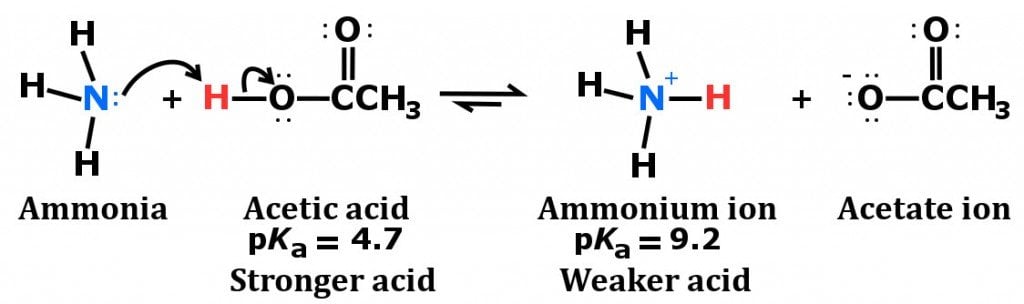
SOLVED: Answer questions a-c about the Bronsted acid-base reaction below uSing the identifying letters A-D below each structure. The pKa's for the acids of interest are: ammonium ion (pKa-9.3). and acetic acid (

The pK(a) of acetic acid and pK(b) of ammonium hydroxide are 4.76 and 4.75 respectively. Calculate the pH of ammonium acetate solution.
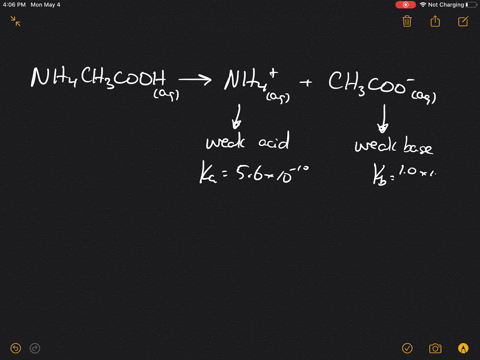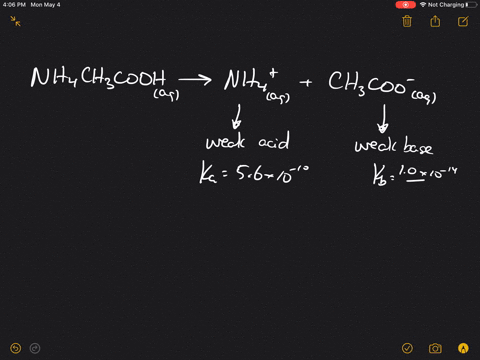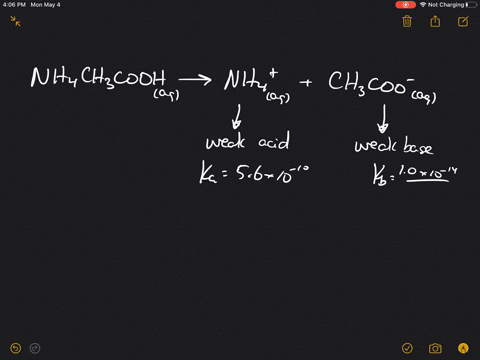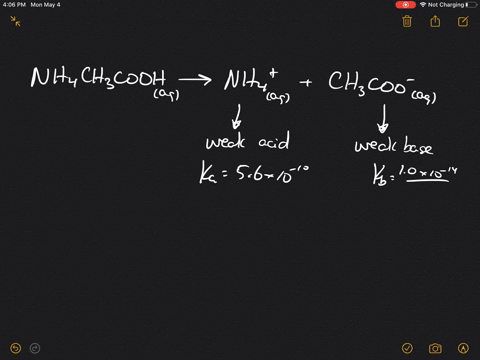
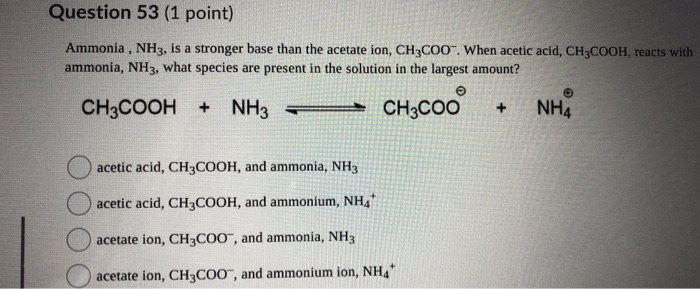
SOLVED:Which of the following salts produces an acidic solution in water: ammonium acetate, ammonium nitrate, or sodium formate?